Quality Management Group
Our Quality Management Group (QMG) is responsible for the planning, organisation and direction of a quality management system within all divisions of The Doctors Laboratory in accordance with appropriate accreditation and regulatory bodies.
The aims of the Quality Management Group are to:
- Meet the needs of relevant evolving regulatory standards (e.g. ISO 15189 Medical Laboratories).
- Ensure that sufficient resources and management time are devoted to quality.
- Put all areas of TDL and TDL Genetics under central quality management control.
- Formalise External Quality Assessment management procedures.
Quality Management Group Operations
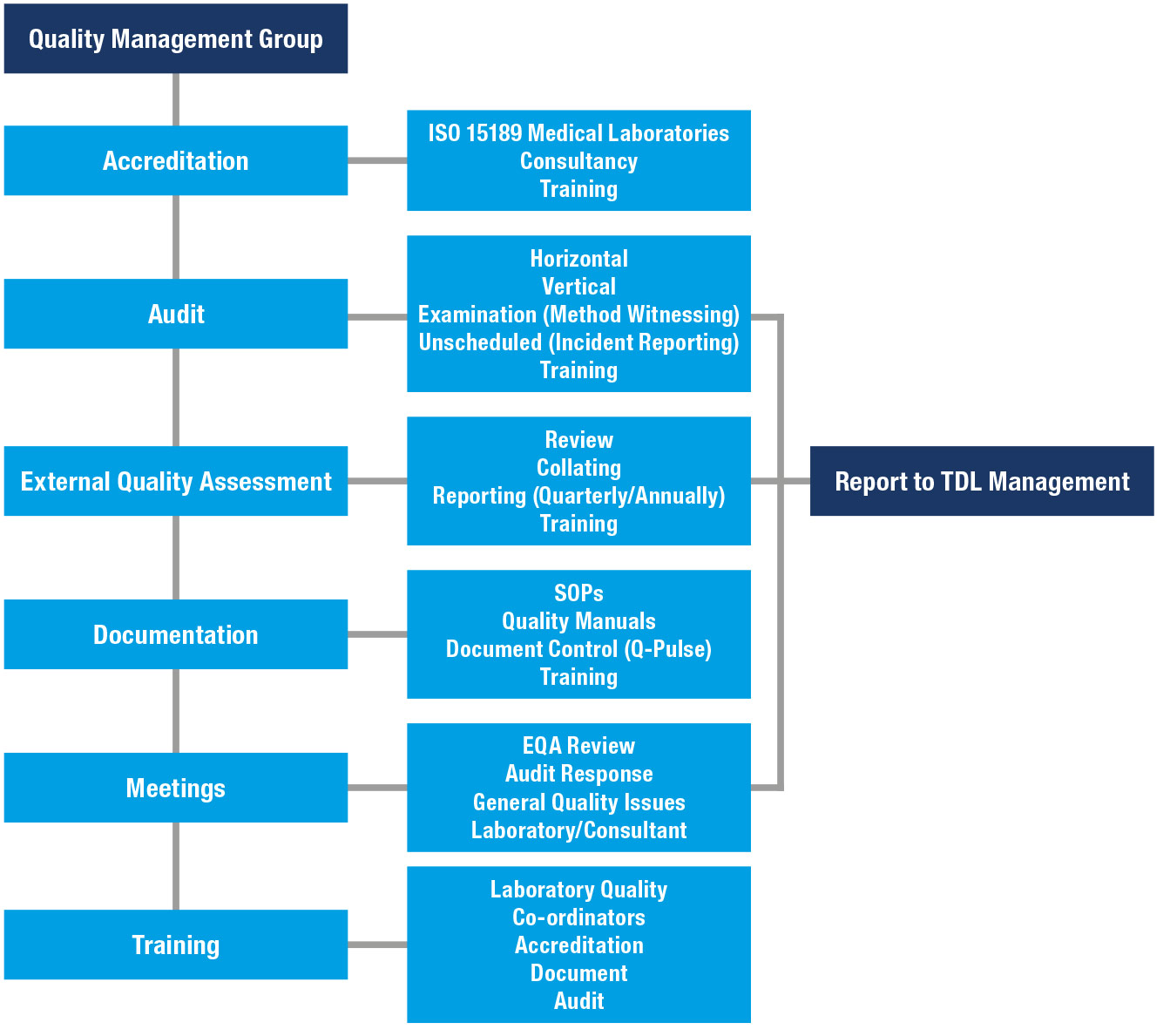
The Quality Management Group’s operations include, but are not limited to:
Review of quality matters
The TDL and TDL Genetics Quality Management Group (QMG) meets on a regular basis, at the appropriate site, to review all quality matters pertaining to the various constituent laboratories of TDL, and TDL Genetics.
The meetings are normally held between the senior members of the QMG and the Laboratory Quality Co-ordinators and/or Quality representatives and can also include senior TDL and TDL Genetics group management and consultants. Frequent meetings between the senior members of the QMG are also held.
Quality matters reviewed include External Quality Assessment (EQA) returns and associated analytical problems, quality standards and training, internal quality assurance audits and their responses and overall quality system management and documentation.
EQA reviews are a key function of the QMG meetings. The purpose is to review and assess the performance of all EQA by reviewing the returns. Any performance that is considered to be outside the limits set by the EQA scheme organisers is quickly tackled and subsequently monitored.
The laboratories receive and initially review EQA returns after which they are discussed at the QMG meeting. The department's consultant also reviews the returns. Results from the return are summarised within a quarterly report for the TDL and TDL Genetics board of directors.
Out of limits EQA data is reported immediately to the senior members of the QMG who monitor remedial action and report promptly to the TDL and TDL Genetics senior management if necessary.
Assuring the quality of data and ensuring compliance
Regular audits and inspections of the laboratory functions and all areas of the Quality Systems are used to assure the quality of the data generated and to ensure compliance to the required standards (e.g. ISO 15189 Medical Laboratories).
The procedures use audit schedules to ensure that all required areas and facilities are inspected on a regular basis and that inspection findings are followed up after a suitable time period. They provide a mechanism whereby audit findings can be reported quickly to senior management.
The areas inspected within the laboratories include, but are not limited to, personnel, facilities, equipment, reagents, standard operating procedures (SOPs), raw data, archives and results. Procedures also exist to inspect and monitor general compliance of the various accreditations and regulations the Organisation subscribes to.
Preparation and maintenance of quality manuals and documentation
The QMG are responsible for the preparation and maintenance of quality manuals for all sites and quality management documentation (QMG SOPs etc.) for all areas within the organisation.
Most quality accreditation bodies require the production and maintenance of a quality manual, which describes the quality management system of a laboratory and includes the quality policy arrangements for its implementation. Quality manuals for all sites should be available to all personnel within the laboratories and should be reviewed regularly, updated as required, and any changes communicated to all personnel concerned.
Advice, interpretation and training
The QMG provides expert advice and interpretation on quality matters and relevant standards to all areas within the organisation. Where necessary, the Group is also responsible for the provision of appropriate Quality training to all members of the QMG and staff at all sites within the organisation.
Definitions
Quality Assurance (QA) is the use of monitoring techniques, including inspections and audits, to promote confidence in laboratory results, to assure the quality of the data generated and to ensure compliance with accreditations bodies such as ISO 15189 Medical Laboratories.
Quality Control (QC) is the use of techniques to reduce discord and discrepancy in results of measurement of the same quantity in the same material
External Quality Assessment (EQA) is the process whereby a laboratory’s analytical performance is assessed with respect to its peers using analytical material obtained from an external provider such as a United Kingdom National External Quality Assessment Scheme (UKNEQAS).
